Paragonix Technologies Raises $24M in Series B Led by Signet Healthcare Partners
Leader in organ transplant technologies secures institutional growth capital to drive organ preservation innovation and commercial expansion
Paragonix Technologies, a leading organ transplant company, has secured $24 million in Series B funding, led by Signet Healthcare Partners. This growth investment will bolster Paragonix Technologies’ commercial footprint, including its ongoing extension of clinical service offerings and inventory expansion, and will enable the Company to further advance transplant innovations.
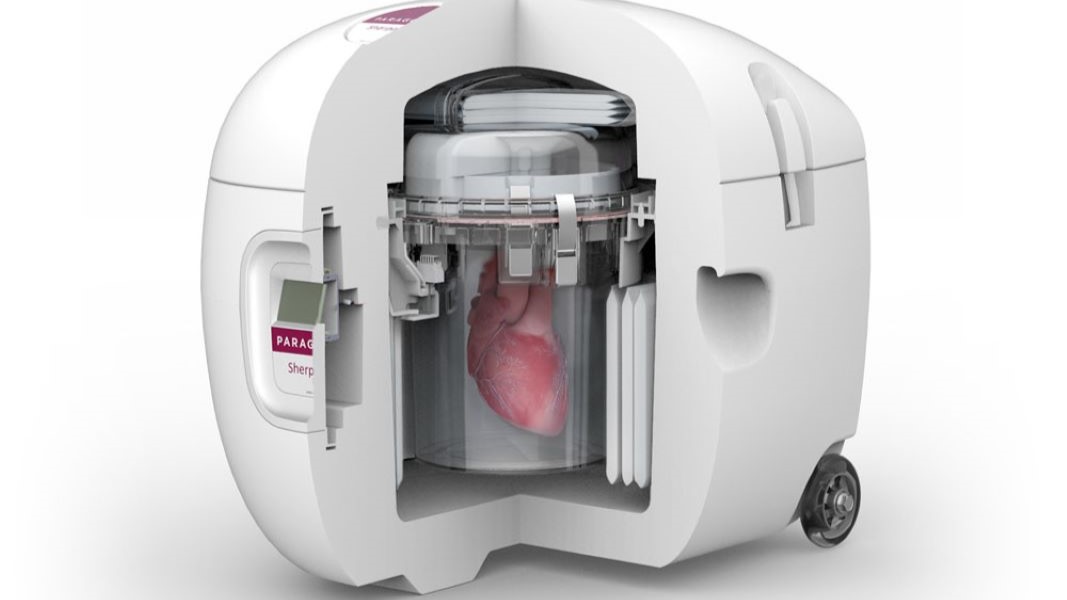
Paragonix Technologies, a leading developer and manufacturer in the organ transplant industry, offers three FDA-cleared and CE-marked devices: Paragonix SherpaPak, LUNGguard Donor Lung Preservation System, and the LIVERguard System. All three devices utilize first-of-its-kind preservation methods that present a significant improvement over traditional ice and cooler practices still used today. Paragonix devices combine clinically-proven, stable preservation technology with a comprehensive clinical support network, augmented by the largest clinical registries of organ preservation data worldwide. They also incorporate digital tracking and communication technologies to provide complete control over organ tracking, monitoring, and reporting.
“The future of Paragonix is full of promise and excitement. Our focus remains on creating the best possible product to help more people receive a second chance at life in a safe and effective manner,” said Dr. Lisa Anderson, CEO of Paragonix Technologies. “This growth investment arrives at a critical moment as we strive to meet rising demands for our organ preservation products. We are excited to rapidly advance our innovation pipeline to enhance our ability to safely preserve and transport as many donor organs as possible. We are thrilled to have Signet as our partner in accelerating growth and achieving our ambitious long-term strategic objectives, and we look forward to leveraging their vast experience in the medical device market.”
Ashley Friedman, Managing Director at Signet Healthcare Partners, stated, “At Signet, we seek to invest in highly innovative and fast-growing healthcare companies. Paragonix has rapidly become a leader in the organ transplant market and is having a significant impact on the future of transplant medicine. We are impressed by their commercial and clinical achievements to date and are excited to partner with their dedicated and versatile team during the next phase of growth.”
In 2022, over 1 in 5 thoracic donor organs transplanted in the U.S. were preserved and transported using a Paragonix device. The Paragonix SherpaPak System is the most commonly utilized FDA-cleared and CE-marked medical device for U.S. heart preservation and transport. According to 2022 transplant data, 19 out of the 30 largest U.S. heart transplant programs, as well as over 80 global transplant programs, rely on Paragonix devices to safely preserve, track, and transport critical donor organs to their intended recipients.
Gunderson Dettmer acted as legal counsel for Paragonix Technologies. Sheppard, Mullin, Richter & Hampton LLP and Knobbe Martens acted as legal counsel for Signet Healthcare Partners. Terms of the transaction were not disclosed.






