MD Anderson and ARTIDIS Announce Strategic Alliance to Advance Nanomechanical Technology Platform for Optimizing Cancer Treatment



FDA-approved CAR T-cell therapy fuels investor funding of the first-ever t-cell cryopreservation startup Cell Vault, the world’s first t-cell…

Innovative program expands to 26 facilities throughout California to better support providers during COVID-19 Blue Shield of California announced the…
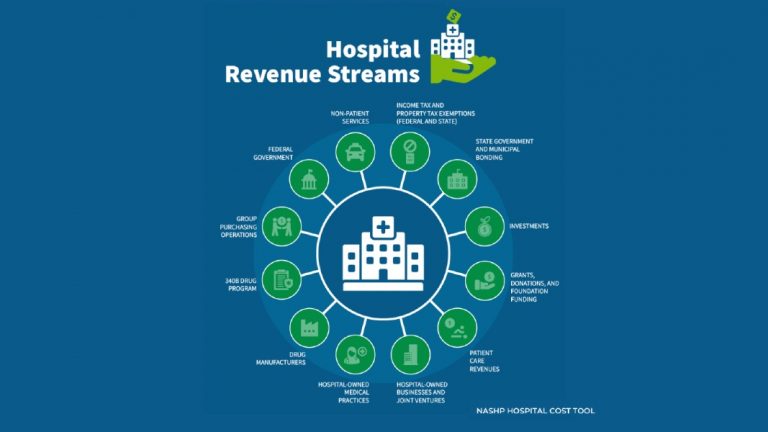
Data will be critical to informing effective cost-containment policy options for states Hospitals provide critical care for many…